
Lithium ion battery, today are not limited to smart phones and laptops only, they forms a critical part of the modern power infrastructure, supporting the renewable energy plant to large scale power grids and high reliability substations. Earlier the power system was depended upon diesel generators, lead acid battery banks for backup power and powering the control circuits and protection system relays, utilities and industries are now transitioning towards the lithium ion battery solution because of its high efficiency, faster response with high energy density and longer service life supporting 3000 charging cycles.
Table of Contents
Construction of Lithium ion battery
A lithium ion battery is built out of the following components for safe storing and efficient release of energy.
Anode construction
The anode or the negative electrode is made up of graphite or carbon based material because graphite has a layered structure, which allows reversible intercalation of lithium ions, the ions can leave and enter repeatedly during the battery operation without damaging the material. Also, graphite exhibits very less change in volume during the cycling with high electrical conductivity.
Fine carbon powders (carbon black, acetylene black or carbon nanotube) are mixed with the graphite to form conductive network, which reduces the internal resistance, improve electron transport and ensure uniform current distribution inside the electrode.
A polymer-based binder most commonly polyvinylidene fluoride PVDF is used to hold the electrode composite together and bind it to the copper current collector which is typically of 7-15 micrometers. The binder provides the mechanical cohesion and adhesion of the electrode to current collector. It may be noted that copper is chosen as the current collector because it does not alloy with the lithium at operating potentials, maintain excellent conductivity while being stable during repeated cycling.
Cathode construction
The cathode or the positive electrode of the lithium ion battery acts as a source of lithium ions and operational voltage. The base of the cathode is basically a thin aluminum foil of 10-20 micrometers, which collects and supplies the electrons during charging and discharging process. The aluminum current collector also provides the mechanical support to the active materials of the electrode while being chemically stable during high potentials.
The active material of the cathode is coated on the current collector. The common industrial chemistry for the active material are
| Chemistry | Chemical Formula | Key Characteristics |
| LCO (Lithium Cobalt Oxide) | LiCoO₂ | High energy density, consumer electronics |
| NMC (Nickel Manganese Cobalt) | LiNiMnCoO₂ | EVs, grid storage, balanced performance |
| NCA (Nickel Cobalt Aluminum) | LiNiCoAlO₂ | EVs, high energy |
| LFP (Lithium Iron Phosphate) | LiFePO₄ | High thermal stability, long life |
| LMO (Lithium Manganese Oxide) | LiMn₂O₄ | High power, moderate energy |
This material determines the voltage, capacity and energy density of the battery. Since the lithium metal oxides are poor electrical conductors, conductive carbon like carbon black, graphite powder, carbon nanotubes are added to the cathode to create electron conduction network.
The powdered cathode mixture is bonded to the aluminum current collector using the polymer binder commonly polyvinylidene fluoride.
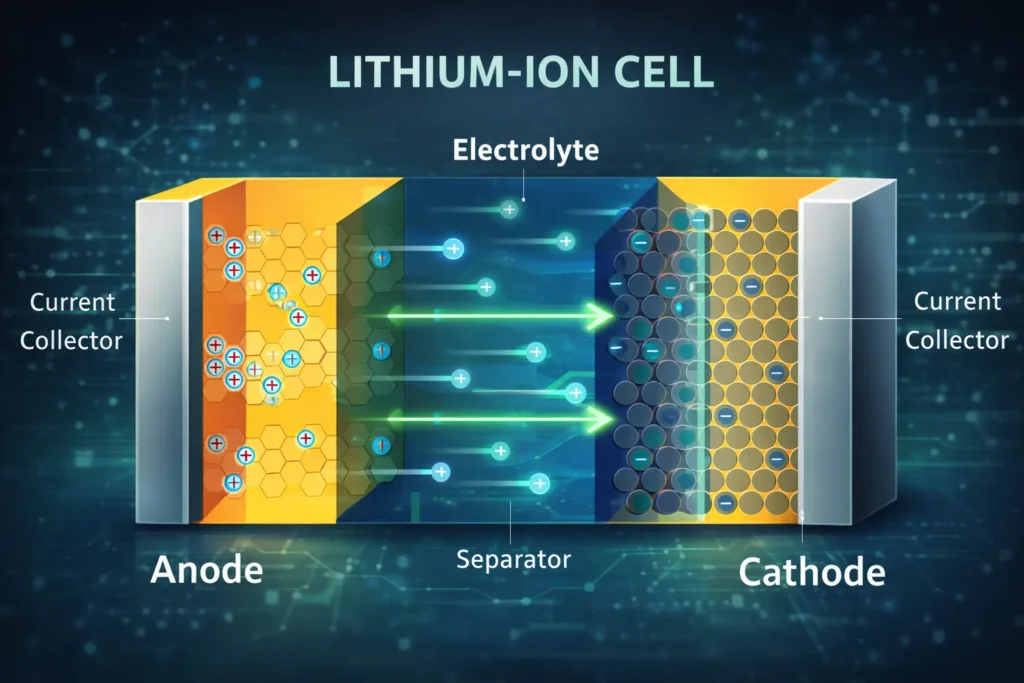
Electrolyte
It is the ion conducting medium, which enables the lithium ions to move between the anode and cathode. The core components of the electrolyte used in Lithium ion battery is Lithium salt, organic solvent and functional additive.
The most common lithium salt for a lithium ion battery is Lithium hexafluorophosphate (LiPF6) which has high dissociation into Li+ and Pf6+ and is stable in typical operating range with low moisture tolerance. The Lithium salt releases the Li+ ions which moves between the anode and the cathode. The concentration of the salt directly determines the ionic conductivity of the electrolyte, internal resistance and power capability of the lithium ion battery. Alternative salts for specialized cells include LiBF4, LiTFSI, LiFSI.
The organic solvent in which lithium salts are dissolved functions as the transporting medium of the lithium ions. The lithium salts dissolves in the solvent which maintains the salt dissociation and prevent ion recombination. It enables the electrolyte to partially decompose on the anode surface and form a solid electrolyte interphase layer. This layer enables the Li+ ions to pass and blocks further electrolyte decomposition, preventing direct reaction between the graphite and electrolyte.
The solvent provides the ion mobility as it determines the viscosity of the electrolyte, speed of the lithium ions and performance at low and high temperature. Low viscosity solvents enable faster ion transportation.
| Solvent | Role |
| Ethylene Carbonate (EC) | Forms stable SEI layer |
| Dimethyl Carbonate (DMC) | Lowers viscosity |
| Diethyl Carbonate (DEC) | Improves low-temperature performance |
| Ethyl Methyl Carbonate (EMC) | Enhances ionic mobility |
The electrolyte additives are performance and safety enhancers which are added in small quantities (0.5-5%). Certain additives suppress the gas formation, stabilizes the solid electrolyte interphase, reduce flammability and improve thermal performance, providing overcharge protection. Common additives are Vinylene carbonate (VC) and Fluoroethylene carbonate (FEC).
Separator
Between the anode and the cathode, there is a separator, which is thin porous membrane made of polypropylene or polyethylene or composite materials with a typical porosity of 40% by volume, which allows the lithium ions to pass and prevents the electrical short circuits. It prevents direct contact between the electrodes while maintaining the ionic conduction. Advanced separators include ceramic coatings which maintains structural integrity up to 700 °C.
Cell architecture
Lithium ion battery are manufactured in cylindrical, prismatic and pouch cell configurations. Many cylindrical cells uses jelly roll design, where anode, cathode and separator layers are made into a spiral and inserted into metal casing. Each cell is inserted in to protective casing with pressure relief vents and a positive temperature coefficient device, which controls abnormal conditions such as overheating and overcurrent.

| Parameter | Cylindrical Cell | Prismatic Cell | Pouch Cell |
| Physical shape | Round metal can | Rectangular metal can | Flat flexible laminated pouch |
| Mechanical enclosure | Rigid steel or aluminum can | Rigid aluminum casing | Polymer–aluminum laminated film |
| Energy density (volumetric) | Moderate | High | Highest |
| Energy density (gravimetric) | Moderate | High | Very high |
| Thermal behavior | Excellent heat dissipation due to metal shell | Good heat dissipation | Limited heat dissipation; needs external thermal management |
| Mechanical robustness | Very high; strong casing | High; good structural integrity | Low; requires external mechanical support |
| Safety containment | Excellent pressure containment | Good pressure containment | Poor inherent containment; relies on pack design |
| Manufacturing complexity | Mature, automated, low defect rate | More complex | Most complex |
| Cell-to-cell consistency | Very high | High | Moderate |
| Module design flexibility | Limited due to fixed geometry | Moderate | Very high |
| Cost per kWh | Lowest (mass production advantage) | Medium | Highest |
Working principle of Lithium ion battery
A lithium ion battery works by moving the decomposed lithium ion in the electrolyte back and forth between the anode and cathode. At the same time the electron flows through the external circuit creating the usable electric power. The process is reversible allowing the battery to be charged and discharged many times.
Charging
During charging of the lithium ion battery, the battery is connected to a charger, which pushes the lithium ions out of the cathode. The ions travel in the electrolyte and enters the anode and are stored in the graphite layers. At the same time, electrons move towards the anode via external circuit.
Reaction at the cathode is oxidation reaction, where the cathode releases the lithium ions. Taking LiCoO₂ as the active material,
LiCoO₂ → Li₁₋ₓCoO₂ + xLi⁺ + x e⁻
Reaction at the anode, where the lithium ions are stored in the anode by intercalation into graphite. This reaction is reduction.
xLi⁺ + x e⁻ + C₆ → LiₓC₆
The lithium ions enters the graphite layers and electrons arriving through the external circuit to the anode combines with the ions and thus chemical energy is stored in the graphite structure.
Overall charging reaction
LiCoO₂ + C₆ → Li₁₋ₓCoO₂ + LiₓC₆
X represents the fraction of lithium atoms which are transferred from cathode to anode, it shows the state of charge of the battery. When x is 0, it means battery is fully discharged and anode contains no lithium. As x increases, lithium moves from cathode to anode. X is never equal to 1 because if all lithium is removed from the cathode, its structure will collapse. The value of x typically ranges from 0.5-0.8 depending on the chemistry.
Discharging
When the battery is powering a device or a system, the lithium ion leaves the anode and travels back to the cathode via the electrolyte and at the same time, electrons flows from the anode towards the cathode via the external circuit powering the connected device or system.
The reactions at anode and cathode are reversed of that during charging along with the overall discharging equation which is also reversed.
Li₁₋ₓCoO₂ + LiₓC₆ → LiCoO₂ + C₆
Failure modes of Lithium ion battery
The lithium ion battery degrades through electrochemical, mechanical means that gradually leads to capacity drop. Most lithium ion battery degrades progressively and seldom shows sudden failure issues. Common modes are
SEI layer growth
The solid electrolyte layer formed at the anode during initial charging thickens with each charging cycle and consumes active lithium. Unstable growth of this layer adds to the internal resistance of the battery. It results in fading of the capacity, higher heating and reduced charge acceptance. High temperature, high charging voltage, fast charging and electrolyte decomposition triggers the growth of this layer.
Lithium plating on anode
When charging of the battery is too fast and temperature is low, lithium gets deposited in metallic form on the anode surface instead of getting between the crystal lattice of graphite. It results in loss of active lithium, formation of tree like branching crystal structures called dendrites, higher risks of short circuit and capacity loss.
Cathode structural degradation
Repeated charging and discharging causes lithium insertion and removal which causes micro cracking of the cathode particle, weakens the grain boundaries and introduces localized stress. It results in more decomposition of the electrolyte as fresh internal surface becomes exposed to the electrolyte.
At high potential and elevated temperatures, cobalt, nickel and manganese ions dissolve from the cathode surface into the electrolyte and these ions migrates to the anode and deposits on the solid electrolyte layer and catalyses the electrolyte decomposition, results in internal resistance rise, loss of active material and increase in gas generation.
Electrolytic decomposition
At elevated temperature, high voltages and contamination, the electrolyte breaks down into salt and solvent which accelerates the gas formation, swelling of cells, solid electrolytic layer instability and induce safety risks.
Separator degradation
Excessive heat causes the polymer separator to shrink, collapsing the pores and reducing the ionic conductivity. It increases the risks of thermal runaway, internal short-circuit and high internal resistance.
This article is a part of the Energy storage and reactive power compensation page, where other articles related to the topic are discussed in details.
Why is Lithium used?
Lithium is used because it is extremely light, highly reactive and capable of moving fast inside the battery. These makes the battery store more energy inside a light weight package, delivering high efficiency.
Why is the process stable and long lasting?
The solid electrolyte interface layer formed at the anode during early operation only allows the lithium ions to pass while restricting any unwanted chemical reaction between the active material and the electrolyte making the process stable and ensuring high battery life.
What happens to lithium ion battery under heavy load?
Under heavy load, the movement of the ions speeds up, internal temperature rises along with internal resistance. The battery management system monitors this and adjusts the operation to keep the battery in safe limits.
What is the basic principle of lithium-ion battery?
While discharging of the battery, the lithium ions moves from the anode towards the cathode via the electrolyte and at the same time the electrons moves from the anode towards the cathode via the external circuit delivering power to the connected load. The reverse happens during the charging operation.

