
Lead acid battery is a mature rechargeable electrochemical energy storage technology, which was first used in the year 1859. Lead acid battery finds its application in many industrial and stationary backup power environment like substation, where cost, reliability and high surge capability are the priority. In the power system, the lead acid battery serves as a DC auxiliary source for the protection relay, control circuits and UPS loads. Despite of the modern chemistries, the lead acid battery continues to be chosen because of its history, predictable performance, failure modes and established engineering practice.
Table of Contents
Working principle of lead acid battery
A lead acid battery is basically an electrochemical energy conversion system whose performance is governed by the reaction kinetics, mass transport, internal resistance and thermal behavior.
The lead acid battery operates by converting the chemical energy into electrical energy through a reversible electrochemical reaction between lead, lead dioxide and sulfuric acid.
Electrochemical process
The lead acid battery contains:
- Positive plate: Lead dioxide (PbO2)
- Negative Plate (Pb)
- Electrolyte: Dilute sulfuric acid (H2SO4)
When the external circuit is closed, the discharge process begins.
At the negative plate,
Pb + SO42− → PbSO4 + 2e−
Electrons released travels through the external circuit and delivers the electrical power.
At the positive plate or cathode, the reaction that occurs is
PbO2 + SO42− + 4H++ 2e− → PbSO4 + 2H2O
The sulfuric acid (electrolyte) is consumed and water is produced, which lowers the electrolyte density.
Thus, the overall cell reaction becomes
PbO2 + Pb + 2H2SO4 → 2PbSO4 + 2H2O
This reaction produces an electromotive force which is approximately equal to 2.0-2.1 volts per cell.
During the charging process, when external DC power is applied, the reaction reverses to
2PbSO4 + 2H2O → PbO2 + Pb + 2H2SO4
During the charging, the lead sulfate reconverts into lead dioxide (PbO2) and sponge lead (Pb), and the sulfuric acid concentration increases. Thus, the electrical energy is converted back into chemical potential energy which is stored within the plates and electrolyte.
Ion Transport in electrolyte
During the discharge stage, the sulphate ions (SO42-) and hydrogen ions (H+) moves through the sulfuric acid electrolyte essentially to sustain the electrochemical reactions at the plates. This conduction of the ions in the electrolyte directly controls the reaction rate, current output and internal resistance. As the discharge continues, the sulfuric acid is consumed and replaced by water which reduces the ionic conductivity and slows down the ion transport.
If the ionic conductivity is low, it results in poor transport of the ions, and the reaction rate decreases and vice-versa. The low ionic conduction makes the current output limited and high conductivity results in higher current output as the current output is directly proportional to the ionic conductivity. However, the internal resistance varies inversely with the ionic conductivity.
Electron Transport in the external circuit
The electrons released at the negative plate flows through the plate material and external circuit to the positive plate producing the electrical power. It is the conductivity of the plate grids, active material and external connection which decides the voltage drop and power delivery.
Reaction kinetics
At the interface of the plates and electrolyte, the reaction occurs at a finite rate which is controlled by temperature, acid concentration, surface area of active material and state of charge.
At high temperature, molecular activity increases, which reduces activation energy barrier, increases ionic mobility in the electrolyte and enhances the charge transfer at the interface. However, excessive temperature accelerates aging and corrosion of the grid.
Higher acid concentration increases ionic availability, thereby reducing the concentration polarization, improving ionic conductivity, while the low acid concentration slows the reaction rate and increases the internal resistance causing higher voltage drop.
The reaction takes place at the exposed surface of the active material and hence higher surface area provides more reaction sites, lowers the current density per site and polarization losses while increasing the total reaction rate. With low surface area of the active material, the voltage drop is rapid with higher local current density and higher heat generation.
The state of charge determines the amount of reactants available, the electrolytic concentration and the plate composition. At high state of charge, the acid concentration is high, active material is fully available, internal resistance is low and reaction rate is high. At low state of charge, the acid becomes diluted, active material becomes less reactive and the internal resistance rises.
Mass Diffusion
As the discharge continues, the sulfuric acid near the plate gets diluted faster than it can get replenished via diffusion. This generates a concentration gradient in the electrolyte. Because of this, the reaction rate decreases with increase in local polarization and internal resistance.
The combined effect causes the terminal voltage to drop faster during discharge before the active material is fully consumed.
Construction of the Lead acid battery
The construction of the lead acid battery is engineered to ensure efficient electrochemical reaction, high current delivery, mechanical stability, and long service life. Every internal component of the battery puts out specific function that influences the performance of the battery and its safety.
Container
The outer container of the lead acid battery is made up of hard rubber, polypropylene or reinforced plastic which is acid resistant, mechanically strong and has dielectric properties. The cover or the container usually contains opening for terminals and vent plugs.
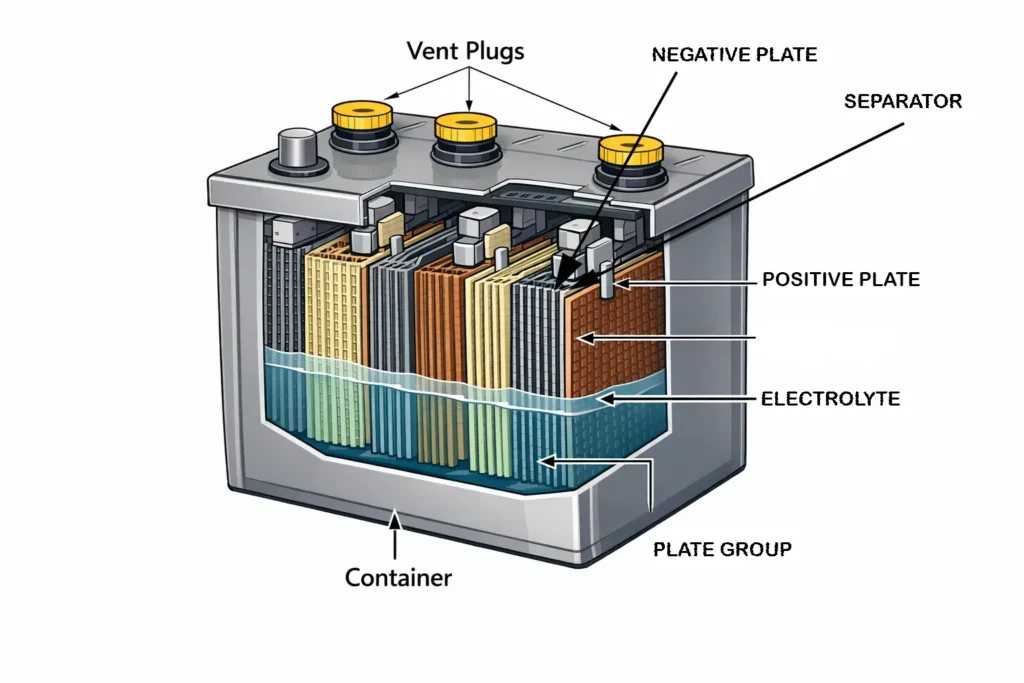
Plates
Each cell of the lead acid battery contains alternating positive and negative plates. The positive plate is made of lead dioxide, which accepts the electrons during discharge and the negative plate is made of spongy lead which supplies the electron during discharge. The plates are mounted on the lead alloy grids which provides the mechanical support, high electrical conductivity and offers resistance to corrosion. Multiple thin plates are used in the construction instead of few thick plates to increase the surface area of the exposed material and the reaction rate.
Separators
Separators are placed between the positive and negative plates which prevents the short circuit and allows the ions to flow through the pores. The separator is usually a microporous polyethylene insulating sheet, which maintains the uniform plate spacing.
Electrolyte
The electrolyte used in a lead acid battery is dilute sulfuric acid (H2SO4) which provides the ions for the electrochemical reaction. When electrons flows through the external circuit, the electrolyte completes the internal circuit by transporting the ions between the positive and negative plates. The concentration of the electrolyte determines the internal resistance and cell voltage. As during discharge, the sulfuric acid is consumed and replaced by water, which reduces the ionic conductivity and increases internal resistance and results in fall of the terminal voltage. The typical specific gravity when the cell is fully charged is usually 1.26 to 1.28.
Plate groups
Plates of same polarity are connected in parallel to form plate groups. The positive and negative groups are alternately stacked and connected to form one cell. Each cell produces approximately 2.1 volts. This is because of the intrinsic electrochemical potential difference between the Pb/PbSO4 reaction (-0.36 V standard potential) and PbO2/PbSO4 reaction (+1.69 V standard potential).
Vent Plugs
Flooded lead acid battery is not a sealed system and is designed with a vent plug for each cell. The vent plug performs functions like release of hydrogen and oxygen gases during charging and thus maintain a safe internal pressure, allows the water replenishment as gas generation consumes the water and the electrolyte level gradually drops. Modern vent plugs incorporates flame arrestor, splash barrier and acid mist trap which reduces the loss of electrolyte and prevents ignition of hydrogen outside the battery.
Failure modes of lead acid battery
Lead acid battery ages and fails through interconnected physical, chemical and mechanical mechanism which is typically driven by charge cycling, charge control, temperature and storage conditions. Understanding these modes of failure will help engineers to plan maintenance, diagnostic and end of life strategies for these battery systems.
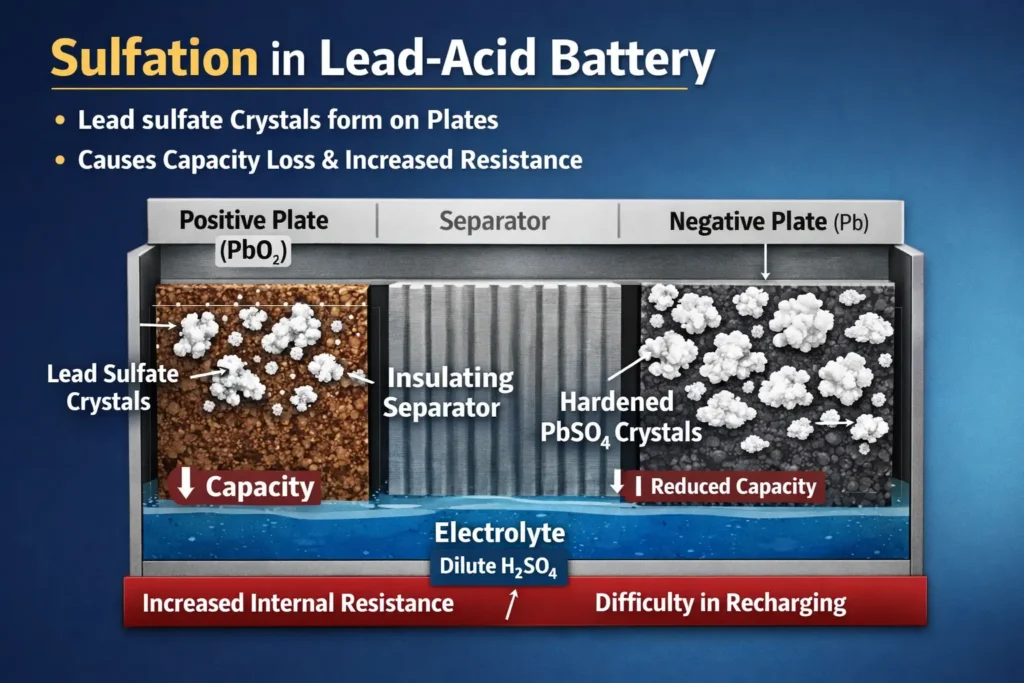
Sulfation
Sulfation refers to formation of lead sulfate on the negative electrode when the battery is left in a discharged state for long. Initial formation is normal chemistry but large, hard and crystalline PbSO4 deposits can never be fully converted during charging. These crystals blocks the pores and active material’s surface area, thereby increasing the internal resistance and reducing the capacity of the battery once the crystals hardens. Therefore maintaining full charge during redundancy dissolves the accumulation of PbSO4 before it hardens.
Positive grid corrosion
The positive grid of the lead acid battery is usually made of lead alloy which gradually oxidizes with time, especially near the high potential during the charging. This corrosion forms resistive PbO2 layer which weakens the conductive grid structure, increases the internal resistance and produces early capacity fading effect. This condition usually occurs at elevated float voltages, high ambient temperature, repeated charge/discharge cycle. Therefore, it is important for engineers to monitor the internal resistance trend and compare cell voltages within a string to detect the grid corrosion onset.
Thermal Runaway
Thermal runaway refers to a self-reinforcing loop of increase in temperature and current that goes out of the battery’s thermal dissipation capacity and ultimately causing catastrophic failure, casing rupture or even fire. This occurs when elevated temperature raises the internal current for a given voltage and higher current generates more heat, increased temperature further lowers the internal resistance and accelerate the reactions. This self-reinforcing loop rapidly escalates if not controlled.
Thermal runaway is triggered by overcharging, poor ventilation, high ambient temperature, imbalanced cell string conditions. The detection clues for this condition are rising float current, inconsistent temperature between the cell and ambient, increasing gas evolution rates. Early detection is crucial to prevent the hazard.
Plate shedding
Plate shedding is a failure mode of the lead acid battery where the active material detaches from the plate because of repeated expansion and contraction under cycling. The loose material then sets at the bottom of the cell and results in reduced active surface area contributing to the capacity loss.
Internal shorts
Hard shorts are caused because of the manufacturing defects or any physical debris bridging the gap of the plates. Soft shorts or dendrites are the metal lead deposits which forms conductive filament through the separators often seen after deep discharge of the lead acid battery. Both types shortens the effective life of the lead acid battery resulting in hotspots.
Predictive indicator of End of Life
Engineers use multiple indicators to estimate the state of health and end of life of lead acid battery.
Increase in internal resistance: Rising internal resistance of a lead acid battery signals grid corrosion, electrolyte loss or sulfation. Significant trend above the designed limits indicates gradual failure.
Reduced capacity: It is measured by periodic discharge test, when the lead acid battery fails to deliver the rated Ah under defined load signals a near end of life.
Imbalanced cell voltage: Repeated low or high voltages within a string indicates localized failure mode like sulfation, shorts or dry out.
Rising float current: It indicates changes in the internal reaction rates, which is an early warning of corrosion or thermal issue.
This article is a part of the Energy storage and reactive power compensation page, where other articles related to the topic are discussed in details.

